













| Still deciding? Get samples of $ ! US$ 1/Piece Request Sample |

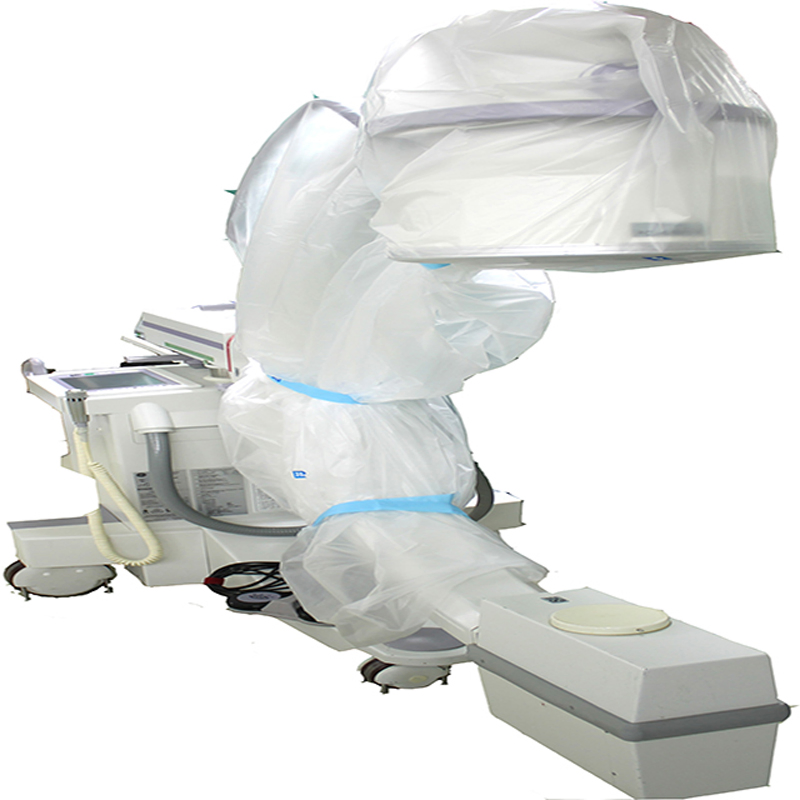
Our C-Arm Covers are designed to provide reliable protection for fluoroscopic and X-ray imaging equipment used in operating theatres. Manufactured from high-quality, latex-free materials, they meet rigorous standards to ensure consistent performance during imaging procedures.
The covers are engineered for simple application and a secure fit, helping to minimise contamination risks while offering a cost-effective solution for maintaining a sterile surgical environment across a wide range of C-arm systems.
The transparent, fluid-resistant material maintains clear visibility to support accuracy, safety and operational efficiency in any clinical setting. Each sterile drape kit is individually packaged and includes straps for securing the cover in place.
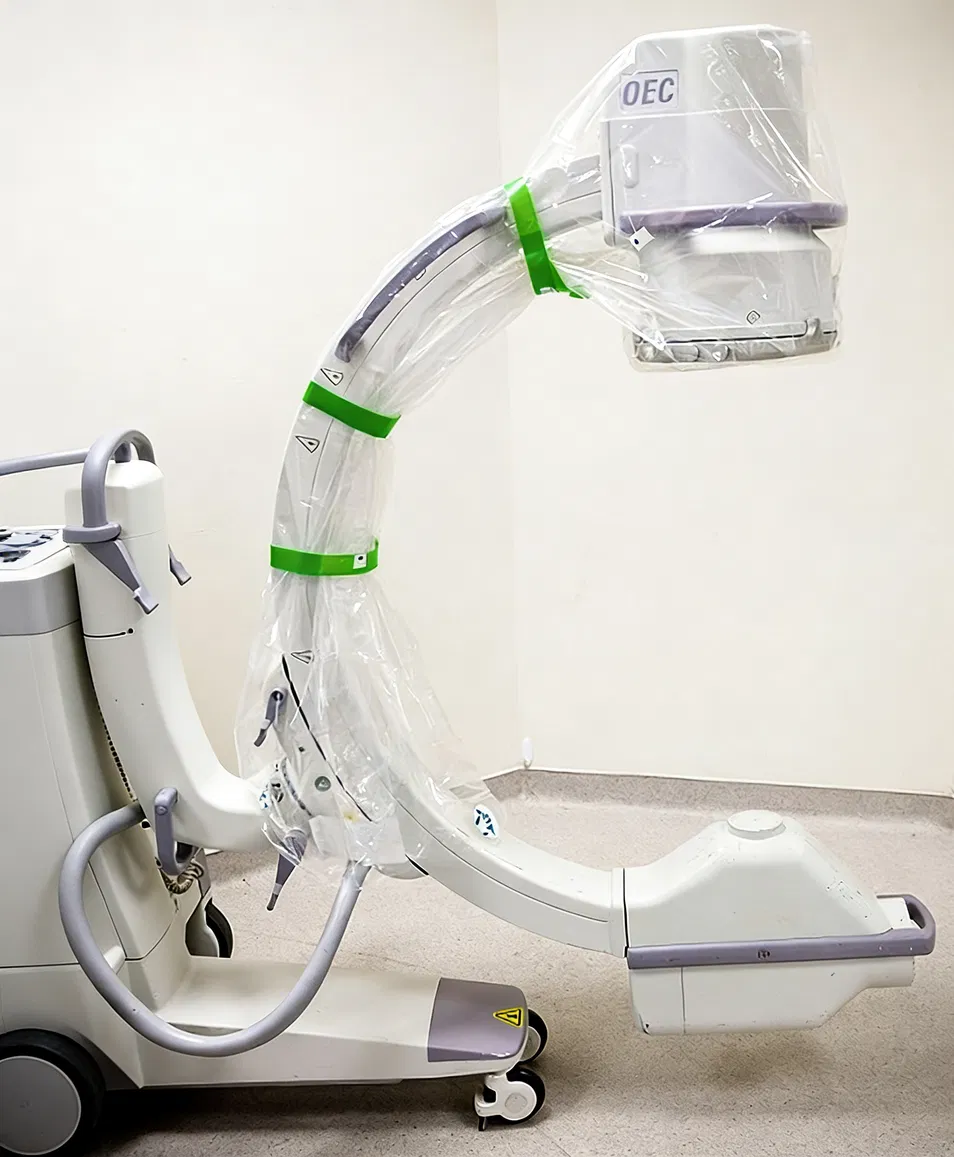
| Ref. | Description | Size | Sterility |
|---|---|---|---|
| C-Arm Cover Kits | Band Bag | 22 in / 56 cm | Sterile |
| C-Arm Cover Kits | C-Arm Cover | 12 x 63 in / 30 x 160 cm | Sterile |
| Clip System Kits | Band Bag | 30 x 26 in / 76 x 66 cm | Sterile |
| Clip System Kits | C-Arm Cover | 12 x 76 in / 30 x 193cm | Sterile |
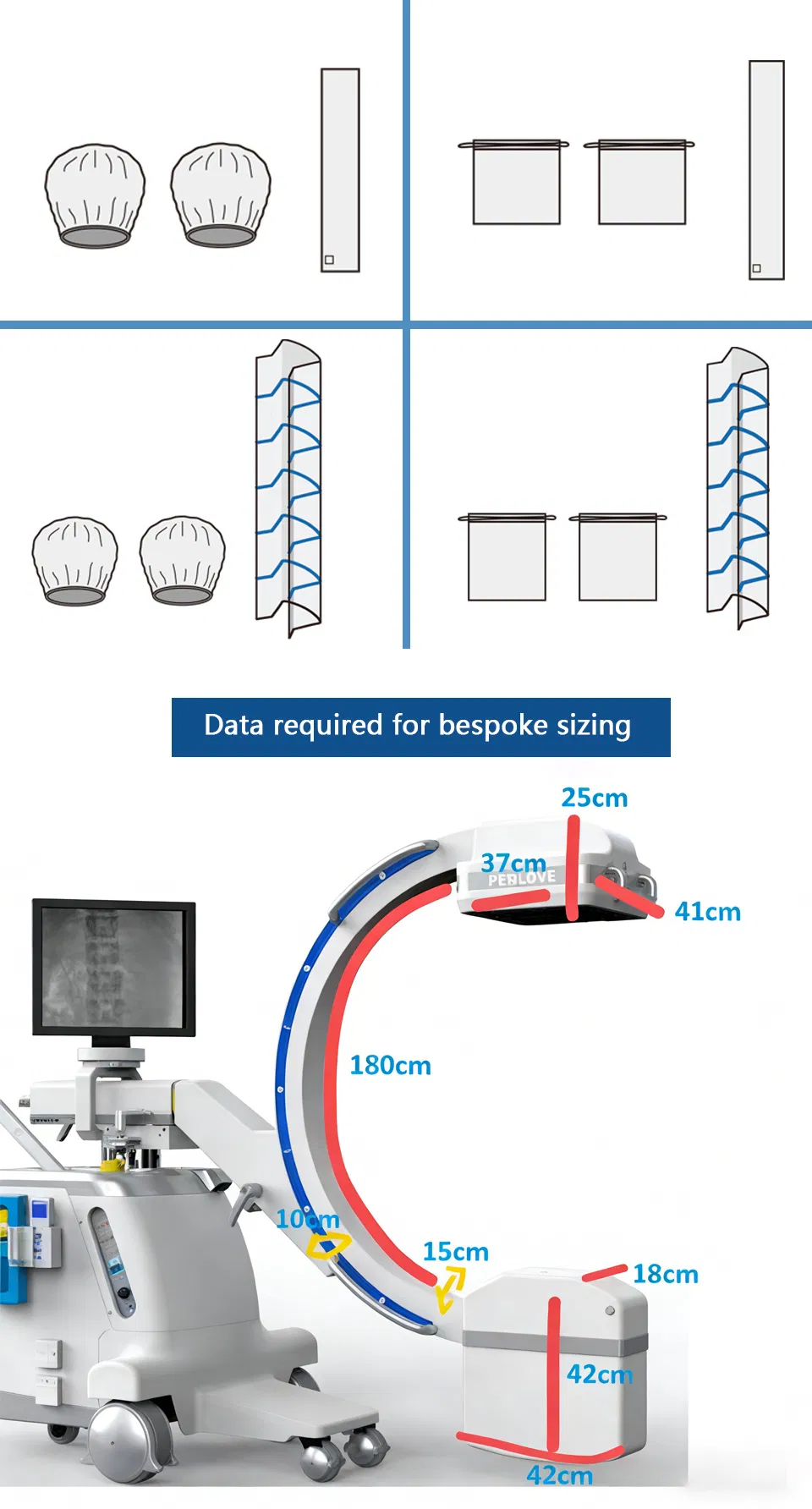


Orthopedics & Trauma
Closed fracture reduction, internal fixation, spine surgery, and real-time fluoroscopic guidance.
Interventional Radiology
Vascular interventions, stent placement, and percutaneous biopsies.
General Surgery & Pain Management
Cholangiography, nerve block localization, and minimally invasive procedures.





We are a One-Stop Medical Consumable Solution Supplier with a 10-year expert R&D team. Our facility features three 100,000-level purification workshops and a professional workforce of over 300 employees.
We specialize in surgical dressings, ultrasound probe covers, and C-arm equipment dressings. Our systems are CE (MDR), ISO, and FDA certified, supporting local registration for customers globally.
 NW Medical
NW Medical