Basic Information
Model NO.
Equipment Drapes
Sterilization
Ethylene Oxide (EO)
Size
Standard / Customized
Safety Standard
GB/T18830-2009
Certification
CE/ISO13485/En13795
To maintain a safe, sterile operating room environment, surgical equipment covers are a surefire way of safeguarding your instruments from infectious bacteria. Medical equipment covers are sterile plastic and can protect the equipment, prolong the service life of medical equipment, improve turnover rates between surgical cases, and avoid contamination.
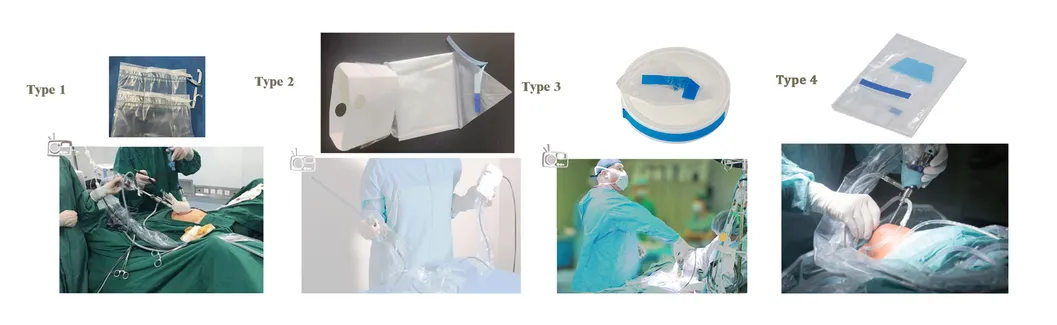
Universal Cover
The product is aseptic packaging, using transparent medical PE blow molding, suitable for the protection of all kinds of machinery and equipment, providing sterile isolation through a protective sleeve.
| Type |
Item Number |
Dimensions |
Packaging |
| Circular Banded |
HTPC30250C |
30*25cm |
50pcs/box; 200pcs/case |
| HTPC50900C |
50*90cm |
50pcs/box; 200pcs/case |
| HTPC13000C |
130*130cm |
50pcs/box; 200pcs/case |
| Rectangle Banded |
HTPC76900R |
76*90cm |
50pcs/box; 200pcs/case |
| HTPC90900R |
90*90cm |
50pcs/box; 200pcs/case |
| HTPC13000R |
130*130cm |
50pcs/box; 200pcs/case |
Light Handle Cover
The light handle cover is a sterile protective cover that covers the shadowless lamp handle, isolating it from the external environment and preventing contamination during operations.
C-ARM Cover
Adopts transparent medical grade PE blown film molding, suitable for protecting the balloon tube and lead plate on DSA machines, preventing blood exudation from polluting the equipment.
Ultrasound Probe Cover
Mainly used for B-ultrasound examinations. Made of polyurethane (TPU) or PE material, offering excellent extensibility and comfort with extremely low allergenicity.
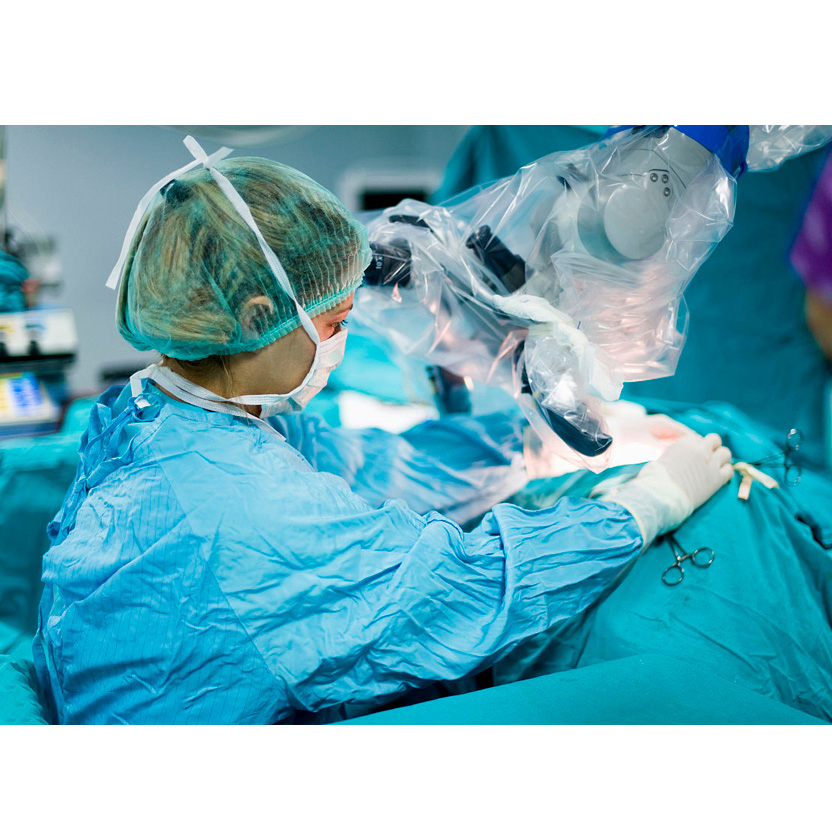
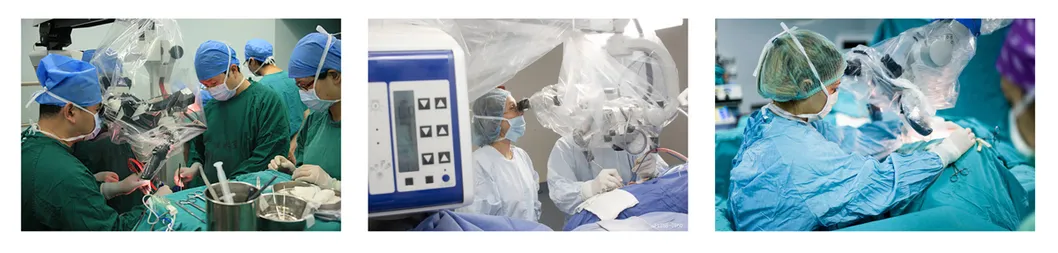
Microscope Cover
Aseptic packaging made of transparent medical grade PE. Designed to be attached directly to the eyepiece and objective lens of operating microscopes to avoid dust and maintain sterility.
Endoscope Cover
Used for the protection of any medical instruments and supplies requiring sterile isolation. Ensures a fully aseptic environment for wires or instruments before operation.
Frequently Asked Questions
Are you a manufacturer or a trading company?
We are an integrated industry and trade company specialized in medical consumables for over 25 years, ensuring both manufacturing quality and supply chain efficiency.
Can I request samples for quality evaluation?
Yes, we are pleased to provide samples for testing and quality evaluation purposes before you place a bulk order.
Do you support OEM or ODM customization?
Absolutely. we support full OEM/ODM services, including customized product dimensions, materials, and specialized packaging or logos.
What certifications do your medical products hold?
Our products are strictly manufactured under quality controls and have passed CE and ISO13485 approvals. Some products also carry FDA certification.
What is the standard lead time for orders?
The typical lead time is 15-20 days. However, for urgent requirements, we can negotiate to shorten the production time based on current capacity.
Do you have quality inspection procedures?
Yes, we perform 100% self-inspection before packaging to ensure every piece meets the required sterile and safety standards.




























 NW Medical
NW Medical